 
(b) In the electron cloud model, the electrons of helium are shown in the variety of locations they would have at different distances from the nucleus over time.Īn atom’s protons and electrons carry electrical charges. 
Although this model is helpful in visualizing atomic structure, in reality, electrons do not travel in fixed orbits, but whiz around the nucleus erratically in a so-called electron cloud.įigure 2.3 Two Models of Atomic Structure (a) In the planetary model, the electrons of helium are shown in fixed orbits, depicted as rings, at a precise distance from the nucleus, somewhat like planets orbiting the sun. In the planetary model, helium’s two electrons are shown circling the nucleus in a fixed orbit depicted as a ring. An electron has about 1/2000th the mass of a proton or neutron.įigure 2.3 shows two models that can help you imagine the structure of an atom-in this case, helium (He). The number of negatively-charged electrons that “spin” around the nucleus at close to the speed of light equals the number of protons. The number of positively-charged protons and non-charged (“neutral”) neutrons, gives mass to the atom, and the number of protons determines the element. 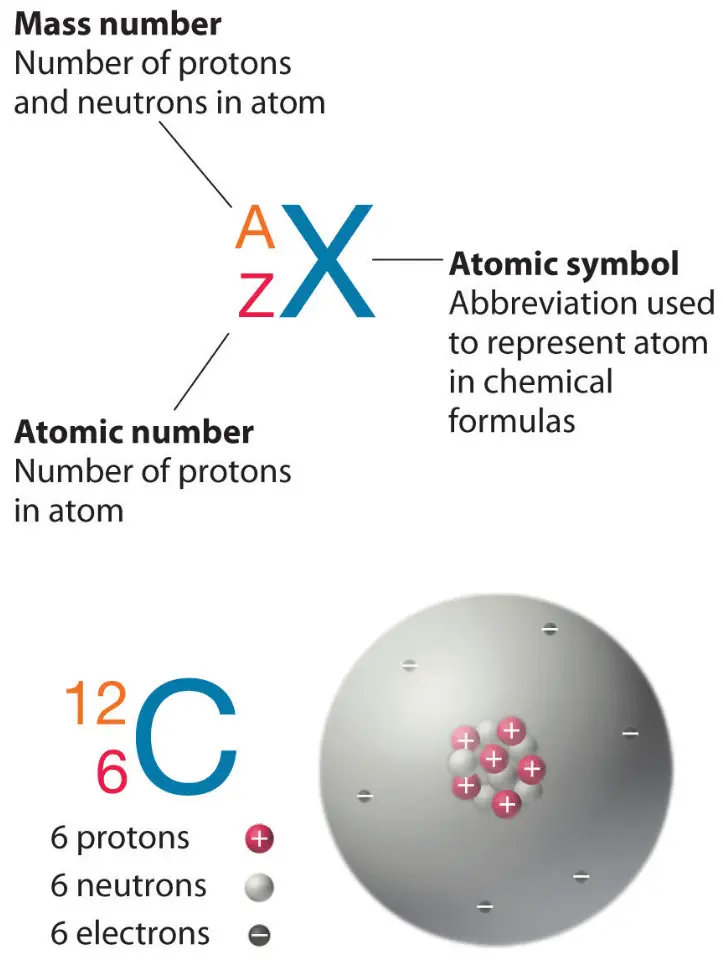
Atomic Structure and EnergyĪtoms are made up of even smaller subatomic particles, three types of which are important: the proton, neutron, and electron. The period at the end of this sentence is millions of atoms wide. As you might guess, atoms are almost unfathomably small. In other words, an atom of hydrogen is a unit of hydrogen-the smallest amount of hydrogen that can exist. But what, exactly, are these “units” of elements? Atoms and Subatomic ParticlesĪn atom is the smallest quantity of an element that retains the unique properties of that element. In glucose, there are always six carbon and six oxygen units for every twelve hydrogen units. Moreover, the elements that make up any given compound always occur in the same relative amounts. It is always composed of the same three elements: carbon, hydrogen, and oxygen. For example, the compound glucose is an important body fuel. A compound is a substance composed of two or more elements joined by chemical bonds. All the elements in your body are derived from the foods you eat and the air you breathe.įigure 2.2 Elements of the Human Body The main elements that compose the human body are shown from most abundant to least abundant. Each element’s name can be replaced by a one- or two-letter symbol you will become familiar with some of these during this course. The elements in the human body are shown in Figure 2.2, beginning with the most abundant: oxygen (O), carbon (C), hydrogen (H), and nitrogen (N). Some other elements you might be familiar with are oxygen, sodium, and iron. 
The elemental calcium in cheese, therefore, is the same as the calcium that forms your bones. Among these is calcium, which, because it is an element, cannot be broken down further. When you consume dairy products your digestive system breaks down the food into components small enough to cross into the bloodstream. Calcium is essential to the human body it is absorbed and used for a number of processes, including strengthening bones. A familiar example of an element that you must take in is calcium (Ca). While your body can assemble many of the chemical compounds needed for life from their constituent elements, it cannot make elements. An element is a pure substance that is distinguished from all other matter by the fact that it cannot be created or broken down by ordinary chemical means. Elements and CompoundsĪll matter in the natural world is composed of one or more of the 92 fundamental substances called elements. A piece of cheese that weighs a pound on Earth weighs only a few ounces on the moon. In other words, weight is variable, and is influenced by gravity. An object of a certain mass weighs less on the moon, for example, than it does on Earth because the gravity of the moon is less than that of Earth. Where gravity strongly pulls on an object’s mass its weight is greater than it is where gravity is less strong. An object’s weight, on the other hand, is its mass as affected by the pull of gravity. An object’s mass is the amount of matter contained in the object, and the object’s mass is the same whether that object is on Earth or in the zero-gravity environment of outer space. An object’s mass and its weight are related concepts, but not quite the same. Scientists define matter as anything that occupies space and has mass. The substance of the universe-from a grain of sand to a star-is called matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
